nader - Aug 28, 2010 11:58 am - Voted 10/10
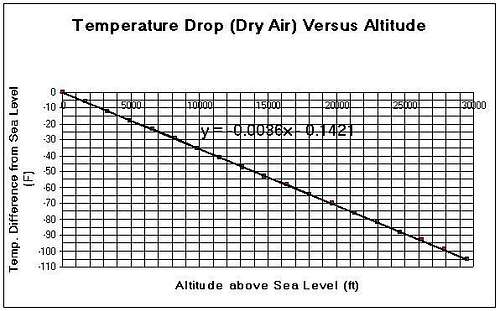
Great ArticleI was still in high school when I got my first (old style) altimeter. I became very interested in finding a formula that would calculate the barometric pressure as a function of elevation. I came up with various formulas that gave an approximate answer. This is a formula that I came up with myself:
Pressure=Square root of (0.01664 hXh+1067089)-0.109h
Where h is in meters and Pressure in milibars
For example for 1000 m, P=932 mb
It wasn't until I went to college when I came across this formula that gives the exact number:
P=(e) to the power of (-0.000125h)
where e is the natural log function
h in meters and P in atm
nader - Aug 28, 2010 3:48 pm - Voted 10/10
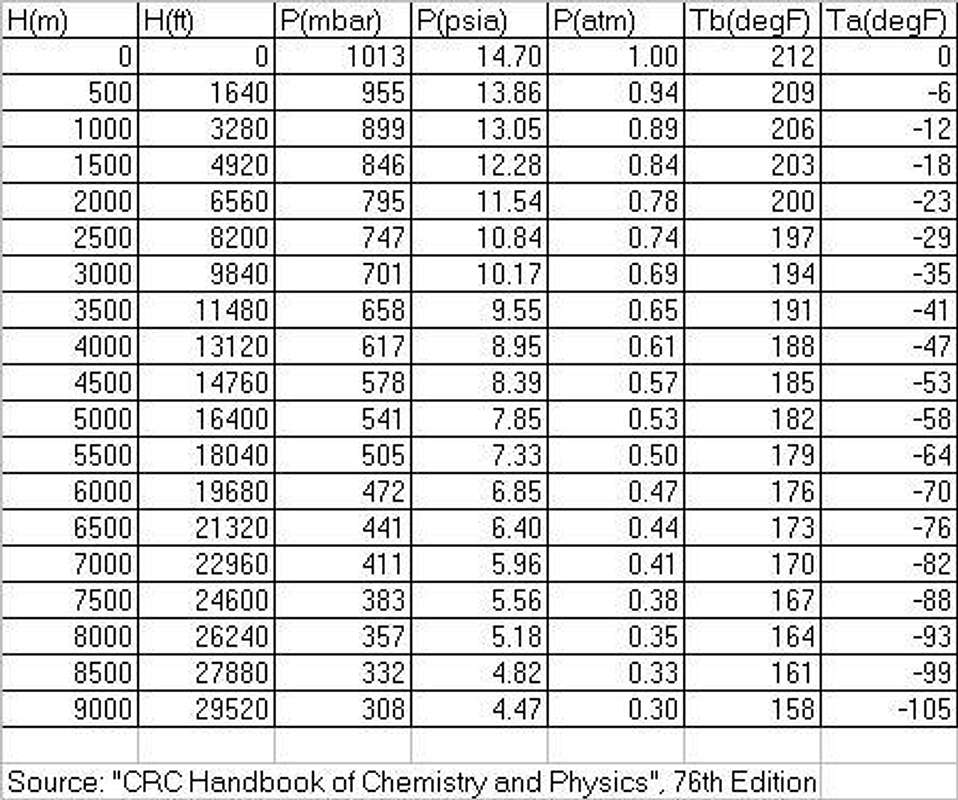
Re: Great ArticleLooking at the numbers in your chart, they seem to be a little off. To begin with, at sea level, air pressure is 76 cm mercury. With a density of 13.6 grams/cm3 for mercury, the pressure becomes 13.6X76=1033 mb. I am not sure why your chart says 1013 mb.
Likewise, for 1000 m elevation, both the second formula that I mentioned and my old altimeter show a pressure of 912 mb, your chart says 899.
Bark Eater - Aug 29, 2010 2:44 pm - Hasn't voted
Re: Great ArticleThanks for your comments. As I referenced, the values for the chart are taken from the CRC Handbook of Chemistry and Physics. It's a standard reference text. I appreciate your interest!
I re-checked the reference. The base document the data are taken from is "U.S. Standard Atmosphere, 1976", published by US NOAA, NASA, and the Air Force. It defines the representation as "idealized, steady state, and during a period of moderate solar activity". Also, in the truth of full disclosure of references, the boiling point data aren't directly from the CRC handbook. I took the air pressures and interpolated saturated steam temperature from a steam table. OK, enough techno-speak for me :-)
nader - Aug 29, 2010 6:47 pm - Voted 10/10
Re: Great ArticleThanks for the reply. I did not mean to be nit picky. I especially enjoyed the info about the boiling point of water at various elevations.
Sierra Ledge Rat - Aug 28, 2010 10:29 pm - Voted 10/10
ThanksIt would be a lot easier if the rest of the world switched to Imperial units instead of metric. (:
coldfoot - Sep 1, 2010 1:25 am - Hasn't voted
Latitude dependenceJust to add a subtlety about the lapse rate - it applies in the troposphere where the atmosphere transports energy convectively, and more or less for that reason, the pressure variation with altitude depends on the overall temperature. The standard atmosphere applies around mid-latitudes. Closer to the poles, the atmosphere is colder and the pressure drops off more quickly with altitude. This underlies the idea that the air is thinner at some altitude on Denali than at the same altitude in the mid-latitudes. The sea level pressure is the same in Alaska, but the pressure falls off faster with height.
Some diagrams:
http://apollo.lsc.vsc.edu/classes/met130/notes/chapter8/graphics/500mb_np_eq.free.gif
http://apollo.lsc.vsc.edu/classes/met130/notes/chapter8/isobar_chart.html
Bark Eater - Sep 1, 2010 12:45 pm - Hasn't voted
Re: Latitude dependenceColdfoot, this is a great addition, of which I was unaware. I hope you don't mind that I added your note to the article text. Thanks much!
bdynkin - Sep 10, 2010 10:45 am - Hasn't voted
Latitude dependence: Denali - Bolivia comparisonI climbed both Denali and a couple of 6K summits in Bolivia so your article prompted me to compare the actual atmospheric pressure in these locations. Luckily, there were semi-permanent weather stations on Denali (5700m), Illimani (6265m), and Sajama (6542m) on some years. It took me maybe 2 minutes to find the actual data for these places. Look here if curious:
http://www.iarc.uaf.edu/mt_mckinley/mt_mckinley_weather.php
http://www.geo.umass.edu/climate/bolivia/data.html
My conclusion after a brief look at the data? Well, in May/June it typically feels 500 to 600 meters higher on Denali compared to the Bolivia mountains! Whoa!
Also, there are pretty extreme pressure variations on Denali between high and low pressure weather events. So summiting Denali on a good day versus bad day could be a difference of up to 700 feet of altutude equivalent!
oldandslow - Sep 30, 2010 5:24 pm - Voted 10/10
Orographic PrecipitationA third phenomenon is the increased liklihood that you will be rained or snowed upon. As the wind blows moist air toward the mountains, the air rises, expands and cools, thereby reducing its capacity to hold water. The result is orographic precipitation. How many time have you returned wet and cold from the mountains and found dry pavement on the flatlands? Probably too many if you live on the west side of the Cascades.
Bark Eater - Oct 1, 2010 10:05 am - Hasn't voted
Re: Orthographic PrecipitationYes, orthographic precipitation is a fascinating phenomenon. I've always been struck by ranges like the Cascades and Sierras that are so wet on the side of the prevailing wind and on top and then have a "rain shadow" out of the mountains on the opposite side.
oldandslow - Oct 1, 2010 2:35 pm - Voted 10/10
Rain ShadowsThere are some remarkable rain shadows along the Pacific Coast. The most dramatic that I know of is on the Olympic Penninsula where the western slopes of the Olympic Mountains approach 200 inches of rainfall annually and the town of Sequim on the east side has less than 20 inches. (Do you really mean "orthographic?)
Bark Eater - Oct 13, 2010 8:31 pm - Hasn't voted
Re: Rain ShadowsYes, I've always seen it referred to as "orthographic". Thanks for commenting!
ExcitableBoy - Oct 1, 2013 10:01 am - Hasn't voted
Orographic vs. orthographicI have always heard the phenomenon referred to as orographic precipitation, but a quick Google search revealed both orographic and orthographic are used.









Comments
Post a Comment